The Bern Birth Cohort (BeBiCo) study recruits pregnant women from the 20th week of pregnancy. Participants are followed up after birth until the child is 10 years old with regular visits. We want to understand how the healthy physical development of the child is supported by the mother. How does the mother, through breastfeeding and other care of the child, help a healthy microbiota to develop and the child's immune system and other bodily functions to develop optimally. For this, a lot of clinical data is collected, many questions are asked and many biological samples are obtained.

With the birth of the first baby in the study in May 2020, the BeBiCo (Bern Birth Cohort) adventure began. During the period from 1st of May 2020 to 31st of May 2023, a total of 116 mother-child pairs were recruited. 11 of them withdrew from the study before or shortly after birth, either because the family was overwhelmed with the arrival of an (another) child, because the infant or the mother developed health problems, or because we unfortunately had to exclude them due to a too distant location of residency. At the time of writing, the youngest children of our cohort were still in their mother’s womb. In contrary, some of the oldest children will celebrate their third birthday this year. Thanks to you, we were able to complete >600 visits and collect about 8’000 samples during this period (stool, skin, vaginal and breast milk samples)
The average age of the “BeBiCo-mothers” at the time of enrollment was 35 years, ranging from 26 to 44 years. 58 participants (56%) were pregnant with their first child. 46 participants (41%) already had children. In the case of one participant, we were later allowed to include the second child in the study.
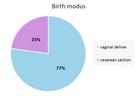
101 participants had given birth: 78 (3⁄4) children were born vaginally while the remaining 23 (1⁄4) were born by cesarean section (Figure 1). This parameter is important for us because it is already known that the mode of birth influences the colonization of the intestinal flora in the first months of life. In our cohort, so far, more boys (55%) than girls (45%) were born.
While a little more than half of the children (54%) were exclusively breastfed during the first four months of life, the remaining children (46%) received formula milk at least once during this period. Very often, infants receive formula milk as supplemental nutrition to breast milk, either because their mother do not have enough milk immediately after birth or because their mother cannot produce enough milk as the weeks go by. Nutrition, especially in the first few months, is considered an important parameter for us because breast milk and formula milk affect the microbiota in different ways. Later, weaning and, to a lesser extent, the transition to solid food mark the change to a microbiota similar to that of an adult.

Trial Steering Committee
(from left to right) Prof. Stephanie Ganal-Vonarburg, Prof. Benjamin Misselwitz, PD Dr. Christiane Sokollik, Prof. Andrew Macpherson, Prof. Daniel Surbek
Operative Mitglieder
Luca Cecchini, Colette Barmaz, Andrina Wick, Nora Rüttimann, Joëlle Zingg, Lea Wahl, Annina Etter, Maria José Coloma Cea, Luana Chanton, Gaëlle Schmutz, Nora-Sophia Staub, Sofia Brunner, Tinevimbo Baricholo, Vanja Tiosavljevic
To study the child's intestinal flora and its development, our study team collects various samples from mother and child at different timepoints in the child's development. The samples collected include the child's stool and skin swabs, which are taken just a few days after birth. Since our gut flora is very dynamic in the first months of life, most samples are collected within the first two years of life, with decreasing frequency. In total, samples will be collected on 11 different days for each child over 10 years of age included in the study. In this way, we hope to record the development of the child's microbiome as accurately as possible with a reasonable amount of effort. Since we suspect that the maternal influence on the child's intestinal flora is crucial, we are also collecting maternal samples, already during pregnancy and beyond. These include maternal stool, vaginal swabs, maternal skin swabs and (as soon as available) maternal milk. By analysing the maternal samples, we hope to draw conclusions about their influence on the development of the child's intestinal flora. Here, the composition of the maternal milk is particularly interesting, as it seems to have a decisive influence on the development of the child's immune system via the intestinal flora. In order to make participation in the study as uncomplicated as possible for the young families, we have decided to collect the samples during home visits. This way, there is as little additional stress as possible in this new intensive phase of life and the new mothers have the opportunity to ask frequent medical questions to our future doctors, who make up the main part of our study team. The maternal samples before birth are taken as part of the routine pregnancy checks in close cooperation with the obstetrics department at Inselspital. The infant and maternal diet strongly influences the composition of the intestinal flora. We record these factors during the study visits by means of detailed nutrition questionnaires. In addition to recording the development of the child's intestinal flora, another aim of our study is to record possible effects of the intestinal flora on the child's health. Therefore, we collect information on the child's growth, development of mental health, as well as on the occurrence of overweight and allergies or other autoimmune phenomena. In order to keep the study participants up to date on the preliminary findings of the study, newsletters are regularly sent to the participants.
The intestinal flora is an important component of our body. According to a study from 2017, the ratio of microorganisms in the body compared to human cells was newly estimated at about 1:1. Our microbiota performs many tasks to keep us healthy: for example, it protects the intestinal barrier from the colonization of pathogenic bacteria, produces essential vitamins, and plays a role in the development and maturation of the immune system.
As long as the child is in the womb, the infant intestine is sterile. The birth marks the beginning of a large-scale bacterial colonization, which will play a role in the development of the child and will shape its immune system. The development of the child’s microbiota is very dynamic and reaches a stable composition, similar to that of an adult, at the age of 2-3 years. Scientific studies show that the first 1’000 days, from fertilization to the end of the second year of life, are fundamental for the development and later well-being of a child. During this time, environmental aspects such as nutrition, hygiene and the place of residence have a formative influence on the intestinal flora and the immune system of the growing child.
As this is a topic that is still mechanistically poorly investigated, our BeBiCo study aims to better understand the maturation of the infant microbiota and the environmental factors (maternal and infant nutrition, mode of birth, home environment, pathogens) that influence this process during the first 10 years of life, with a main focus on the first two years of life.
In a first analysis, we analyzed the microbiota of children who have already reached the first year of life (Figure 2). Each column shows the average microbiota composition at one of the different time points of sample collection. The different colors code for bacteria from a different phylum. All colors together add up to 100%. We can clearly see that the gut flora undergoes a change over the first year of life. While an adult intestinal flora consists largely of bacteria of the
Bacteroidetes (light green) and Firmicutes (pink) species, the infant intestinal flora contains many bacteria that can be assigned to the Actinobacteria (purple) and the Deferribacteres (darker green).. A comprehensive analysis of the stool samples is pending.

Breast milk is the first and a very valuable food for infants and promotes their growth. It contains the three fundamental nutrients (carbohydrates, lipids and proteins), as well as vitamins, growth factors and hormones. Moreover, immunologically active components such as antibodies, cytokines and immune cells are other components of breast milk.
While it has been known for long that maternal antibodies in milk protect the newborn from infections, the effect of these other immunological components in breast milk, such as immune cells, is still largely unknown. And this even though there are more than 1'000 leucocytes (white blood cells) in just one milliliter of milk (Figure 3a).
Via flow cytometric analysis of breast milk, we could already show that more than half of these cells are T lymphocytes, a small fraction B lymphocytes, which produce antibodies, and the rest are natural killer cells and innate immune cells, such as monocytes, phagocytes, and dendritic cells (Figure 3b).
The amount of these cells in breast milk varied highly from mother to mother and there were changes over the time, as we could show here for the frequency of T lymphocytes (Figure 3c). Understanding whether and to what extent differences in the immune cell composition of breast milk influence the development of the child microbiota or immunity will be part of our research in the coming years.

We also determined the amount of certain cytokines in breast milk. For several cytokines (interleukin-15, IL-15, IL-6, and C-X-C motif chemokine 10, CXCL-10, also called interferon gamma-induced protein 10, IP10) as well as several chemokines such as (eotaxin, monocyte chemoattractant protein-1, MCP-1 and MCP-4) we found relevant cytokine or chemokine concentrations in breast milk. Interestingly, cytokine concentrations had been highest at day 10 postpartum, and had decreased again at weeks 10 and 14 (figure 4). The physiological significance of the cytokines for the child is unclear. It is an interesting hypothesis that these cytokines promote the development of the child's intestine and its immune system. We are currently testing this hypothesis in experiments with organoids.

Calprotectin is an inflammatory value in stool, it is contained in or secreted by inflammatory cells (neutrophil granulocytes). Calprotectin in stool is a good indicator of inflammatory activity in the intestine in adults and is used as an activity marker in chronic inflammatory bowel diseases such as Crohn's disease and ulcerative colitis.
In infants and children, calprotectin levels are higher than in adults. This does not necessarily indicate disease activity. High calprotectin levels were also found in the children of BeBiCo immediately after birth and in the first months of life. The inflammation levels had been independent of the mode of birth (section vs. vaginal birth) and independent of the infant diet (exclusive breastfeeding vs. formula + breastfeeding) in initial preliminary studies.
Interestingly, calprotectin levels in the BeBiCo children had then dropped in the first 6 months, while they had remained high in the children of the Zimbabwe comparison cohort (Figure 5). The children of the birth cohort in Zimbabwe live in partly precarious conditions with poor hygiene and frequent infectious diseases. It is possible, therefore, that intestinal inflammation remains high in these children, while it decreases when they grow up under strict hygienic conditions (as in the BeBiCo children). We are currently looking for statistical correlations between the inflammation levels in the stool and clinical data of the participants in both countries (infectious diseases, concomitant diseases, nutrition, hygiene, socio-economic situation).
What significance the different levels of inflammation in the child's gut have for later life is an interesting question. It is possible that the lower inflammation levels reflect the western lifestyle, with little exposure and morbidity from infectious diseases but increased risk of allergies and autoimmune diseases. In contrast, the elevated inflammation levels even at the 6-month time point in the Zimbabwe cohort may reflect the strong engagement of the immune system with the environment, for which our immune system is evolutionarily designed.

